試卷名稱:115年 - [無官方正解]115 國立臺灣大學_ 碩士班招生考試試題:生物化學(A)#137641
年份:115年
科目:研究所、轉學考(插大)◆生物化學
1. Compare the two reaction coordinate diagrams below and select the answer that correctly describes their relationship. In eachcase, the single intermediate is the ES complex.Free energy,
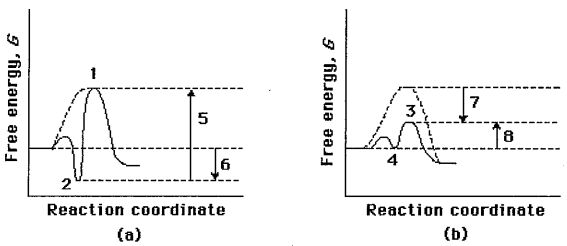
(A) Diagram (a) describes a strict "lock and key" model, whereas diagram (b) describes a transition-state complementarity model.
(B) The activation energy for the catalyzed reaction is #5 in diagram (a) and is #7 in diagram (b).
(C) The activation energy for the uncatalyzed reaction is given by #5 + #6 in diagram (a) and by #7 + #4 in diagram (b).
(D) The contribution of binding energy is given by #5 in diagram (a) and by #7 in diagram (b).
(E) The ES complex is given by #2 in diagram (a) and #3 in diagram (b).